Johnson & Johnson Completes Clinical Study for OTTAVA Robotic Surgical System
Johnson & Johnson's investigational OTTAVA robotic surgical system shows promising safety and performance results in gastric-bypass procedures.
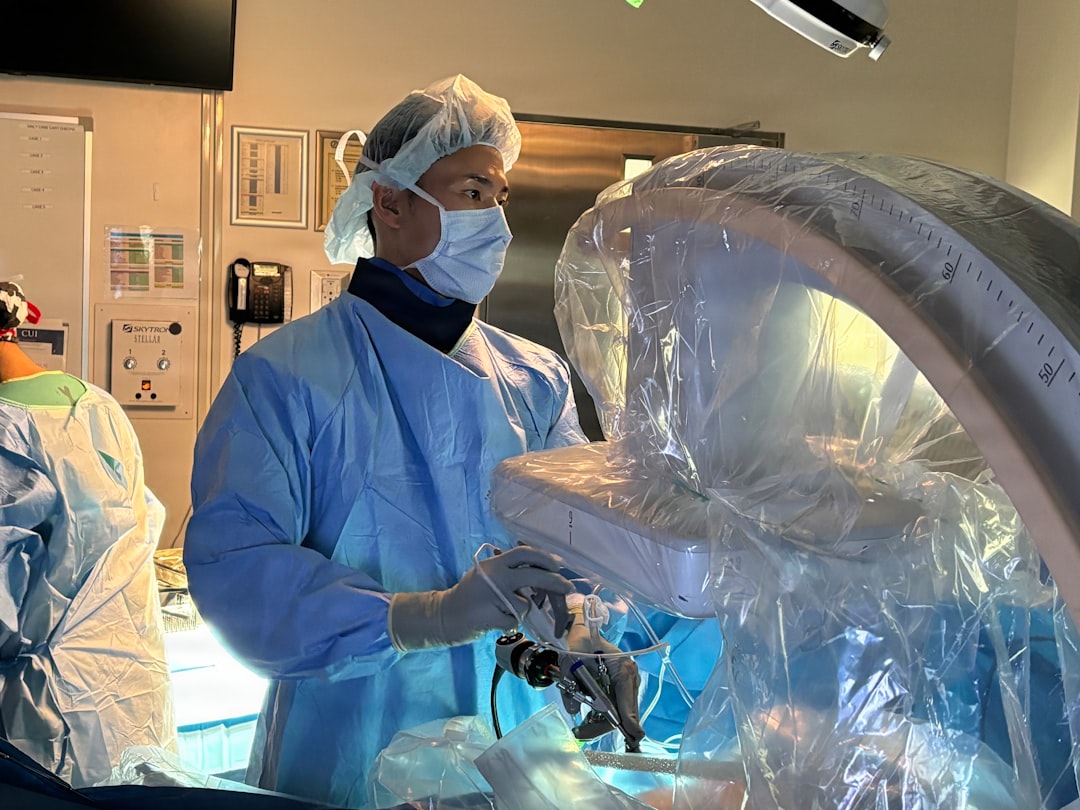
["Johnson & Johnson has announced the completion of a clinical study for its investigational OTTAVA robotic surgical system, designed for procedures such as gastric bypass. The prospective, multicenter clinical study evaluated the safety and performance of the system for gastric-bypass procedures, meeting its primary safety and performance endpoints through 30 days post-procedure. The study involved a 30-patient cohort, with investigators completing all procedures robotically on OTTAVA without conversion to a non-robotic approach.
Average weight loss by 30 days after surgery was 30 lb. (13.6 kg). Dr.
Erik Wilson, chief of minimally invasive and elective general surgery at UTHealth Houston and lead investigator for the clinical study, noted that the data show encouraging evidence regarding the safety and performance of the OTTAVA Robotic Surgical System in Roux-en-Y gastric bypass procedures. The company used this clinical data, together with preclinical testing, to support an application to the U.S. Food and Drug Administration (FDA) for De Novo classification.
J&J is targeting an indication covering multiple procedures in general surgery within the upper abdomen, such as gastric bypass, gastric sleeve, small bowel resection, and hiatal hernia repair. The results were presented at the 2026 American Society for Metabolic and Bariatric Surgery (ASMBS) Annual Meeting. Development of OTTAVA was delayed due to the COVID-19 pandemic, but the FDA granted OTTAVA an investigational device exemption (IDE) in late 2024.
The system completed its first cases a year ago. Johnson & Johnson designed OTTAVA as a multi-specialty soft-tissue surgical robot, enabling a broad range of procedures across multiple specialties. Multiple clinical trials will support these specialties.
The system's novel architecture incorporates four robotic arms integrated into a standard-size surgical table, removing the need for a separate boom or carts. In this study, OTTAVA was installed and used successfully across operating rooms at six participating hospitals, with OR sizes ranging from approximately 243 to 694 sq. ft.
(22.5 to 64.4 sq. m). In five of the six sites, OTTAVA performed procedures in ORs that previously had not been used for robotic surgery, including rooms historically considered challenging for robotic systems due to space constraints.
Dr. Peter Schulam, chief scientific officer for MedTech at Johnson & Johnson, emphasized the importance of collaboration with surgeons and rigorous preclinical work in the development of OTTAVA. He noted that the system's novel architecture supports a differentiated approach, allowing more hospital ORs to accommodate robotics and removing practical barriers to broader adoption."]
Source: The Robot Report